INTRODUCTION
Traditionally the focus of Peru’s mining industry, one of the mainstays of the country’s economy, has been the extraction of copper, gold and other metal ores. However, Peru also offers unexplored potential in the industrial mineral sector. For some years now a research project has been exploring the possibility of industrially exploiting deposits containing feldspar, quartz, clay. Increasing attention is now being focused on industrial minerals, such as zeolites with high ion exchange capacity which are eco-friendly and inexpensive minerals, that find application in a variety of industrial sectors including waste water, gas and steam treatment, in agriculture, in animal feed, excreta and waste amendment. Due to the low cost and high efficiency in the removal of contaminants by adsorption, zeolites are widely used in wastewater treatment 1. Natural zeolites show biological activities due to the ease adsorption and cation exchange. For this reason they are used in medicinal and pharmaceutical processes 2. In 3 it is concluded that zeolites can be used as a food additive due to their unique physical and chemical characteristics.
Heightened environmental awareness among governments and the public in general, calls for a greater commitment by industries and the extractive industry in particular, to reduce pollutant emissions. Pollution in mining areas, known to be associated chiefly with the presence of heavy metals, is caused both by acid mine drainage and by water infiltrating mine waste rock or tailings dumps leaching out the heavy metals contained therein. These elements are transported via runoff and redeposited in some cases at great distances from the pollution source. Thus mine drainage waters need to be treated, removing heavy metals via precipitation in suitable plants or by means of reactive barriers or filters that are able to adsorb the pollutants contained in the water thereby minimizing their diffusion into neighbouring areas. Wang 4 (cited by Ciosek 5 [p. 10]), indicted that natural zeolites are abundant around the world and have been successfully used to solve environmental problems. One of its important properties is the ability to exchange dissolved cations in liquid solutions.
An investigation currently being carried out to address this important issue is aimed at assessing Peru’s mining potential in terms of materials with high ion exchange capacity. This paper presents the first results of an investigation on the Tertiary ignimbritic formations in the Puno area in southern Peru. A mineralogical study carried out on a sample removed from this deposit, where prospection is underway, using diffractometric analysis, and scanning electron microscopy (SEM) revealed the presence of heulandite along with other adsorbent minerals. Technical properties were also determined including CEC, bulk density, specific surface area, and cation exchange capacity of the untreated Na-exchanged material towards NH4+, Pb+2, Cd+2, Cu+2, Zn+2 and Mn+2, singly or in combination. The results of the experimental investigation showed a good cation exchange capacity of the material examined especially towards heavy metals. Furthermore, pretreating the zeolite material with a 2N NaCl solution significantly enhanced the exchange capacity toward all the cations examined. Kragovi 6 (cited by Shi 7 [ p.622], mentioned that the pretreated zeolite improves its adsorption capacity under certain conditions. It must be considered that in a natural zeolite there are pores clogged by the presence of impurities
EXPERIMENTAL
Materials and methods
Material preparation
The sample was reduced in size by open circuit primary crushing using a jaw crusher followed by secondary crushing in a roll crusher operating in closed circuit with a 5mm screen. The crushed product was then classified by screening to 0.7 mm.
In this way two size classes were obtained: -5+0.7 mm and -0.7 mm accounting for around 79% and 21% by weight of the total sample. At this stage of the experimental investigation only the -5+0.7 mm size class was investigated to explore its potential use for the manufacture of filters for purifying contaminated water treating 8.
Characterization
Chemical and mineralogical composition of a sample of this material were determined using a Siemens D5000 diffractometer and LEO EVO 50VP scanning electron microscope (SEM) manufactured by Oxford Instruments equipped with an energy dispersive spectrometer (EDS) microanalyser. At this stage of the experimental investigation only the -5+0.7 mm size class was investigated here to explore its potential use for the manufacture of filters for purifying contaminated water treating.
A Micromeritics Instruments AccuPyc 1330 gas pycnometer was used to determine specific gravity. By means of a Quantachrome Instrument Autosorb-1 automatic surface area and pore size analyser, specific surface area using the BET multipoint method and the following operating conditions; outgas temperature = 150 °C; outgas time = 3 hours; P/P0 ranging from 0.05 to 0.3 (where P0 is the gas pressure used at the test temperature). Cation exchange capacity (CEC), was determined using the non-normalized double-ion exchange method - 1N ammonium acetate solution and 10% potassium chloride solution - proposed by Minato
Na-exchange zeolite
Na-exchange was achieved by placing the zeolite sample in a vessel containing a 2N NaCl solution with solid-to-liquid ratio of 1:4 and leaving at ambient temperature for 10 days modified from 9. The material was then filtered and stove dried at around 40°C.
Reagents
The starting solutions were prepared from the following salts: NH4Cl (99.5%), Pb(CH3COO)2.3H2O (99.0%), 3CdSO4.8H2O (98%), CuCl2.2H2O (99.09%), ZnCl2 anhidro (97.0%) and MnCl2.4H2O (99%), were purchased from Carlo Erba, Italy.
NH4 +, Pb+2, Cd+2, Cu+2, Zn+2 and Mn+2 cation exchange tests
Tests on both, the untreated as well as the Na-exchanged zeolite were carried out placing 5 g of the material (2 g for the tests with the Pb+2 cation) in flasks containing 150 mL solution and agitating at room temperature 10) on a New Brunswick Scientific G 24 environmental incubator shaker (150 rpm).
The calculated amount of cation uptake by the zeolite comes out from the difference between the amount in the starting and final solutions 11),(12. For the metal cations, chemical determinations were done using a spectrometer Perkin Elmer OPTIMA-2100-DV ICP 13),(14. Ammonia was determined using the standard distillation and titration procedure 15.
NH4 +, Zn+2 cation exchange tests varying time
To determine the time required to attain equilibrium conditions preliminary ion exchange tests were performed on the -5+0,7 mm untreated zeolite material following the procedures described above using solutions containing around 1000 mg/L NH4 + and 1000 mg/L Zn+2 respectively.
Tests by varying cation concentrations in the starting solution
These tests were performed following the above described procedures and for a contact time of 24 hours on the untreated and Na-exchanged material and using solutions containing different cation concentrations, examined singly.
The correlation of experimental data with the Freundlich and Langmuir isotherms (Malamis 16, cited by Carbonel17) [p.263], defines the adsorption isotherm as the equation or curve that, in equilibrium condition, relates the concentration of metal adsorbed in the solid phase with the concentration of metal in solution to a specific temperature. The experimental data were correlated using the models proposed by Freundlich and Langmuir:
The Freundlich adsorption isotherm model is given by Ec. (1)
where: X is the amount of cation exchanged per unit mass of zeolite (mg/g); Cf is the equilibrium cation concentration in solution (mg/L); k and n are constants 18.
The Langmuir adsorption isotherm model is given by Ec. (2)
where: X is the amount of cation exchanged per unit mass of zeolite (mg/g); Xm is the maximum amount of cation exchanged per unit mass of zeolite (mg/g); Cf is the equilibrium cation concentration in solution (mg/L); b is Langmuir isotherm constant related with adsorption energy 18. For the determination of adsorption isotherms, non-linear regression analysis was applied, trying to minimize the error between the estimated and experimental data (19).
RESULTS AND DISCUSSION
Mineralogical and Physicochemical Characterization
The analysis by X-ray diffractometer showed mineralogical phases such as; heulandite; mordenite; smectite; quartz; cristobalite and feldspar, evidencing a natural zeolite with the presence of impurities. Figure No. 1, shows the mineralogical species.
Table No. 1, shows the chemical composition determined by X-ray analysis.
Table No 1. Chemical composition of the as received material determined by means of X-ray spectrometry

The chemical analysis indicates that the zeolite contains alumina silicates with a high content of silica and is strongly alkaline.
The following laboratory determinations were performed:
Specific gravity = 2.32 g/cm3, Specific surface area = 33.56 m2/g, Cation exchange capacity (CEC) = 50 meq/100 g. Low specific surface area indicates low porosity due to low zeolitic crystallinity (20).
NH4 +, Zn+2 cation exchange tests by varying time
Cation uptake by the zeolite was calculated as the difference between the amount in the starting and final solutions. Figure No. 3, shows cation uptake for each of the ions examined, expressed as milligrams per gram of zeolite versus contact time.
Figure 3 clearly reveals that after roughly 20 hours in the operating conditions the amount of cation exchanged remains practically constant. Thus, in subsequent characterization tests, in line with experiments reported in the literature, the contact time between the zeolite material and the cation containing solutions was set at 24 hours to ensure the steady-state condition (21).
Tests by varying cation concentrations in the starting solution
Tests were performed following the above described procedures and for a contact time of 24 hours on the untreated and Na-exchanged material and using solutions containing different cation concentrations, examined singly. Figures No. 4, 5 and 6 show the NH4 +, Pb+2, Cd+2, Cu+2, Zn+2 and Mn+2 uptake respectively for the untreated and Na-exchanged material, expressed as milligrams per g of zeolite, and as percentage with respect to the starting solution, versus cation content in the starting solution (mg/L)
Correlation of experimental data with the Freundlich and Langmuir isotherms
Tables No. 2 and 3 show for the untreated and Na-exchanged zeolite material respectively the values of the Freundlich and Langmuir isotherm constants and the corresponding correlation coefficients (R) for the experimental equilibrium values.
Table No. 2. Untreated zeolite material. Values of Freundlich and Langmuir isotherm constants and corresponding correlation coefficients (R) for experimental equilibrium values
Table No 3. Na-exchanged zeolite material. Values of Freundlich and Langmuir isotherm constants and corresponding correlation coefficients (R) for experimental equilibrium values
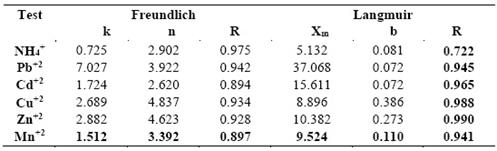
Examination of the results obtained for the ion exchange tests on the untreated material with size class in the -5+0.7 mm range using solutions containing the single cations examined reveals that: The exchange equilibrium relationships are well described by the Freundlich isotherm. It can be seen that the values of R, are close to unity and that the values of n are greater than 2, which indicates a favorable adsorption process on the surface of the zeolite (22).
The selectivity of the zeolitic material towards the cations examined differs in the order: Pb+2> Cd+2> Cu+2> Zn+2> Mn+2> NH4 +, in agreement with the findings reported by other researchers for metallic ions. Zamzow [23] (cited by Kraljevic Pavelic [24, p.2]) aligned the selectivity of clinoptilolite zeolite as follows: Pb+2> Cd+2> Cs+> Cu+2> Co+2> Cr+3 >Zn+2> Ni+2> Hg+2. This depends on the fact that adsorption phenomena are influenced by charge density as well as by hydration energy and ionic radius of the cations.
For the metal ions examined here, exchange capacity decreases with ionic radius, greater exchange capacity corresponding to greater ionic radius (Pb+2). Charge density being equal, the smaller ions (Mn+2) exhibit higher electric charge as they hydrate more readily, encountering greater difficulty in penetrating the zeolite pores to reach the available exchange sites. Note that exchange capacity towards NH4 +, though not negligible, is inferior not only for Pb+2, as occurs for other zeolitic materials, but also to the other metal ions examined. Presumably the significant amounts of clay contained in the zeolitic material tested here contribute to uptake of the metal ions examined.
For the material Na-exchanged with a 2N NaCl solution, the results obtained show that: The metal cation exchange equilibrium relationships are better described by the Langmuir isotherm than by the Freundlich isotherm and denotes that adsorption is multilayer and that there is surface heterogeneity in the adsorbent (25).
This can be explained by the fact that during pretreatment of the zeolite material with a strongly alkaline solution (2N NaCl) the ions present are replaced by Na in the exchange sites. Subsequently, when the Na-exchanged material comes into contact with solutions containing bivalent metal cations, exchange occurs more readily with Na and greater exchange capacity is achieved for lower cation concentrations in the starting solution. Only for NH4 + did the Freundlich isotherm provide a better correlation with the experimental results. For this cation, the selectivity of the zeolite is strong due to the lower hydrated ionic radius that it presents. Petrous [26] (cited by Santiago [27, p. 14]) confirmed that selectivity of clinoptilolite is higher for NH4 + due to its lower hydrated radius ion 3.3 Å. Similarly to the untreated zeolitic material, the Na-exchanged zeolite also exhibits different selectivity in the order: Pb+2> Cd+2> Zn+2> Cu+2> Mn+2> NH4 +, though in this case the positions of Cu+2 and Zn+2 are inverted. Further investigation is warranted to explain this behavior, which can presumably be attributed to changes in the properties of the Na-exchanged material. Under the same operating conditions, the Na-exchanged material proved to be more effective in removing the cations than the untreated material. As an example, for a cation concentration of 1000 mg/L in the feed solution, uptake by the Na-exchanged material was 32%, 55%, 47%, 25%, 53% and 42% higher for NH4 +, Pb+2, Cd+2, Cu+2, Zn+2 and Mn+2 respectively than the untreated material. The improvement of the Na-exchanged removal capacity can be explained by the enrichment of Na+ cations on the natural material and create new available sites and that are accessible for incoming cations [27].
CONCLUSIONS
The mineralogical investigation carried out on a sample of zeolite material, revealed the presence of zeolite minerals such as heulandite and mordenite along with smectite, quartz, cristobalite and feldspar.
Laboratory tests showed the material to exhibit good cation exchange capacity towards the ions examined in the order: Pb+2> Cd+2> Cu+2> Zn+2> Mn+2> NH4 +. Pre-treating the material with a NaCl solution significantly increases the adsorption capacity for all the tested ions. In light of the encouraging results obtained and the need to extend the investigation to representative samples of the area as a whole, it is recommended that prospection be continued so as to determine more accurately the actual size and characteristics of the deposit with a view to its industrial exploitation.












 uBio
uBio